
PDF Viewer
Quality Assurance Service Company of the Year 2026
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
The process of ensuring healthcare quality assurance, for RSL Healthcare Consulting, LLC goes far beyond its definition of services that meet the high standards for excellence, patient safety, and effective outcomes. RSLHCC, founded and led by Randy S. Langsam, a seasoned risk manager/healthcare compliance officer, assists office based surgical facilities in not only meeting, but exceeding state, federal and accrediting agency regulations to be at the highest standard of compliance and patient safety. This is done by guiding them 24/7 and being an integral partner in the process.
Your Partner in Patient Safety

About RSL Healthcare Consulting, LLC

Your Partner in Patient Safety
RSL Healthcare Consulting, LLC (RSLHCC) is dedicated to ensuring patient safety and compliance for office-based surgical facilities. Led by expert Randy S. Langsam, the firm provides personalized, hands-on risk management services that foster trust and elevate operational standards for healthcare providers.
About RSL Healthcare Consulting, LLC

RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
The process of ensuring healthcare quality assurance, for RSL Healthcare Consulting, LLC goes far beyond its definition of services that meet the high standards for excellence, patient safety, and effective outcomes. RSLHCC, founded and led by Randy S. Langsam, a seasoned risk manager/healthcare compliance officer, assists office based su
The process of ensuring healthcare quality assurance, for RSL Healthcare Consulting, LLC goes far beyond its definition of services that meet the high standards for excellence, patient safety, and effective outcomes. RSLHCC, founded and led by Randy S. Langsam, a seasoned risk manager/healthcare compliance officer, assists office based surgical facilities in not only meeting, but exceeding state, federal and accrediting agency regulations to be at the highest standard of compliance and patient safety. This is done by guiding them 24/7 and being an integral partner in the process.

Putting Patient Safety First
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
At the heart of RSL Healthcare Consulting’s work is safety. RSLHCC works with every client to ensure the safety of both patient and staff, and the approach ensures that all necessary risk management measures are in place. We start at the beginning and work from A through ZZZ, as long as it takes, through the policies, equipment, supplies,
At the heart of RSL Healthcare Consulting’s work is safety. RSLHCC works with every client to ensure the safety of both patient and staff, and the approach ensures that all necessary risk management measures are in place. We start at the beginning and work from A through ZZZ, as long as it takes, through the policies, equipment, supplies, medications, sterilization, infection control practices, medical records, credentialing, facility licenses, required signage, trainings and so much more to ensure that every stone is turned and compliance is at 100%. Working this closely with each facility helps to maintain the strict rules and regulations while fostering and enforcing the expected culture and creating a true partnership.
“It’s about working in tandem with the facility and making sure all risk management / healthcare compliance steps are covered for every patient, every single day,” Langsam explains.
By ensuring that everyone, including surgeons, anesthesia providers, surgical support staff, administration and owners are aligned, RSLHCC assists facilities to create smoother operations thus creating greater patient satisfaction, a truer sense of a happy work environment and more effective healthcare delivery.

A Personal Approach to Compliance
RSL Healthcare Consulting, LLC Embodies Healthcare Quality Assurance with its Own Personal Touch!
A Personal Approach to Compliance
What truly sets RSL Healthcare Consulting, LLC apart from other risk management firms is the personal touch. Unlike many companies, especially since Covid, that rely solely on virtual consultations, Langsam and her team visit each client on-site every quarter. This hands-on personal approach allows them to see us, speak to us and ensures
What truly sets RSL Healthcare Consulting, LLC apart from other risk management firms is the personal touch. Unlike many companies, especially since Covid, that rely solely on virtual consultations, Langsam and her team visit each client on-site every quarter. This hands-on personal approach allows them to see us, speak to us and ensures compliance at a higher level. It absolutely helps to build a stronger relationships with the staff. When it’s virtual, you see what they want you to see and you speak to who they want you to speak to.
From the sterilization processors to the surgical assistants and especially the surgeons; we review infection control practices, sterilization processes, surgical regulations and so much more! Some of the best times are the times debating the rules with the surgeons when they POP-IN to speak with us. That does not happen virtually! The familiarity of RSLHCC being on site with these visits enables staff to feel more comfortable, to ask questions, and to seek advice when needed, leading to more open communication and improved care for patients.
At the Forefront of Providing Healthcare Compliance Services
Recognized as a TOP 10 2023 Company in Healthcare Compliance
https://www.healthcarebusinessreview.com/rsl-healthcare-consHouse Bill 1471 Set to take effect on 7/1/23
See our COVID-19 Page - Stop The Spread to Learn More






Help To STOP THE SPREAD!!!
COVID-19 Policies and Fast Facts Manuals and training...Now Available
RSL Healthcare Consulting, LLC and our affiliate RSL Employee Compliance
are proud to present our
COVID-19 Infection Control and Fast Facts Manuals to Healthcare Facilities and Businesses,
which includes a LIVE face-time training as well as Q&A Session for your employees!
Please contact us to see how we can assist your company in helping to STOP THE SPREAD
We can be reached at: Info@RSLHCC.com


Proud to Announce
RSL Healthcare Consulting, LLC is proud to announce our partnership with the Florida Society of Plastic Surgeons. Established in 1956, the Florida Society of Plastic Surgeons has been helping to make safe, high-quality plastic surgery available in the state of Florida for more than half a century.
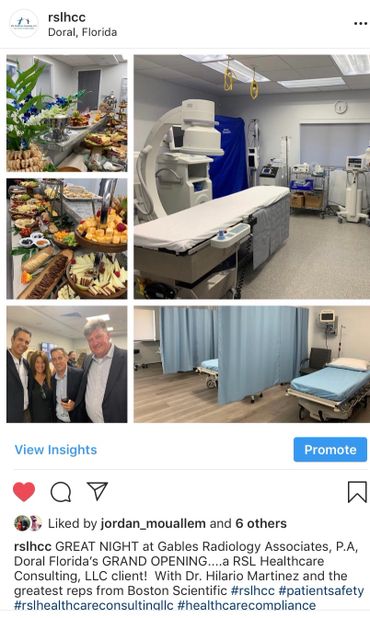
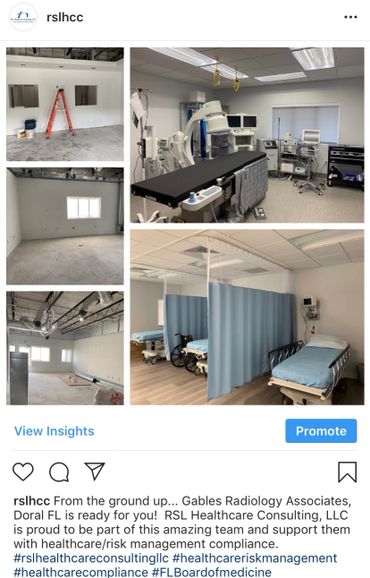
All Hands On Deck!!! Everyone Has A Role in Patient Safety!












Subscribe
Sign up to learn more about RSL Healthcare Consulting. LLC and how we can help your practice
Contact Us
Visit or Call to Schedule an Appointment
Please contact us directly with any questions, comments, or scheduling inquiries you may have.
RSL Healthcare Consulting, LLC
Hours
Monday - Friday: 9AM–5PM
Saturday - Sunday: Closed